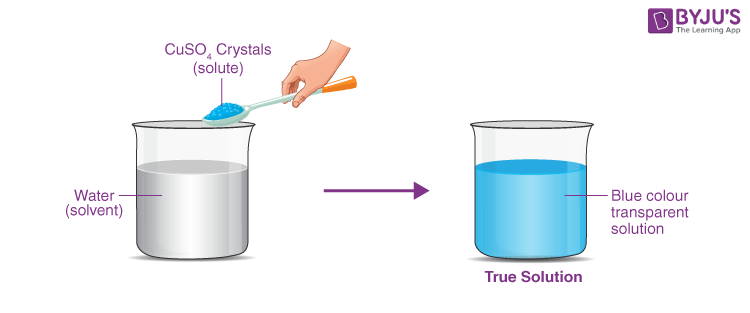
CBSE Class 9 Chemistry Practicals and Experiments on Preparation of a True Solution of Common Salt, Sugar and Alum

Homogeneous Solution of Water and Salt and Heterogeneous Mixture of Water and Sand in Glass Beakers. Stock Illustration - Illustration of icon, clipart: 119285559

Simple saltwater solution could help reduce early symptoms of coronavirus, research suggests | London Evening Standard | Evening Standard
Is a solution of salt and water considered to be a chemical substance. Are some mixtures considered to be chemical substances and some not, or are all mixtures considered to be not
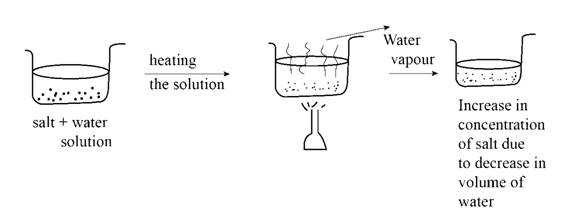
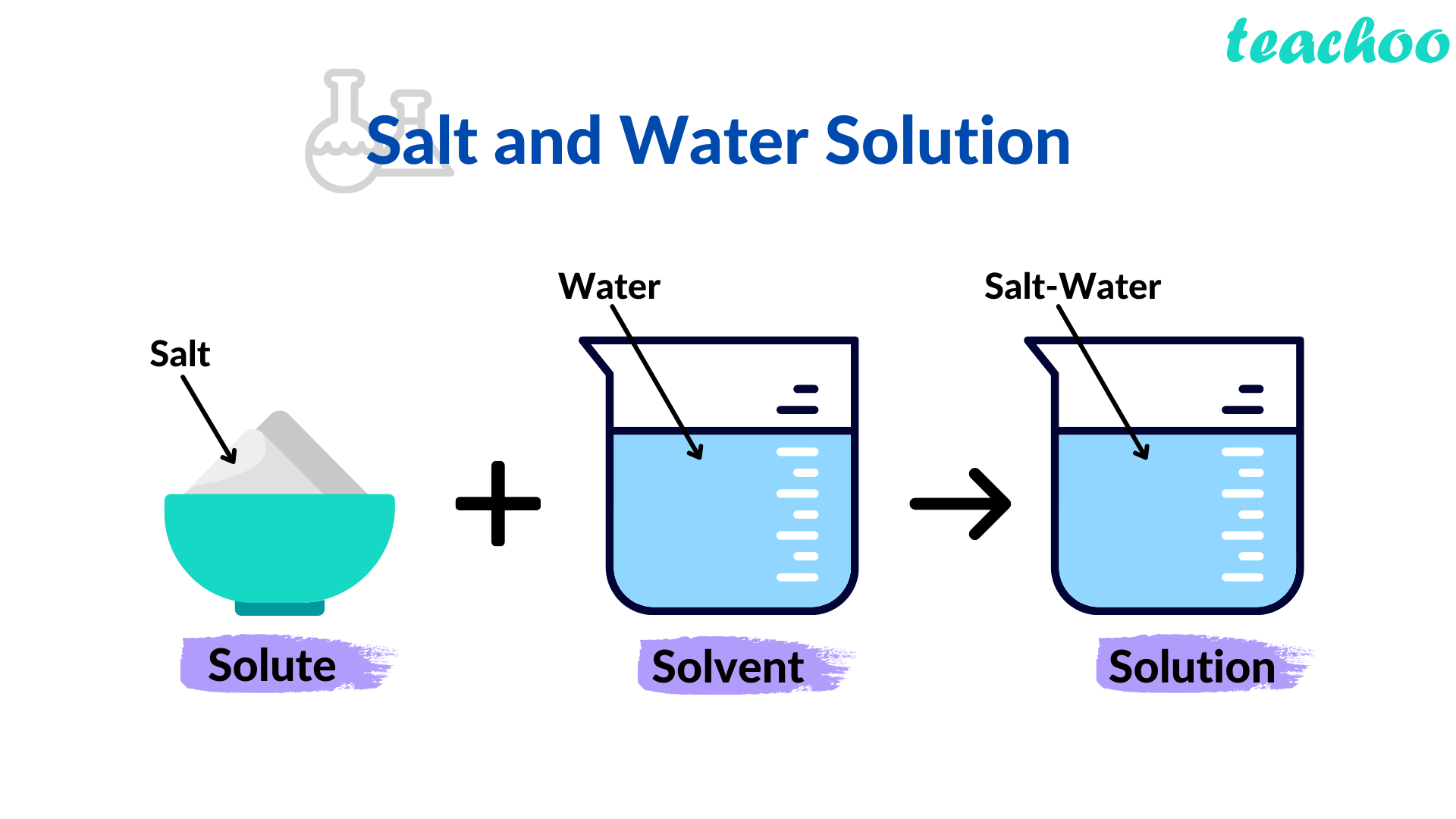



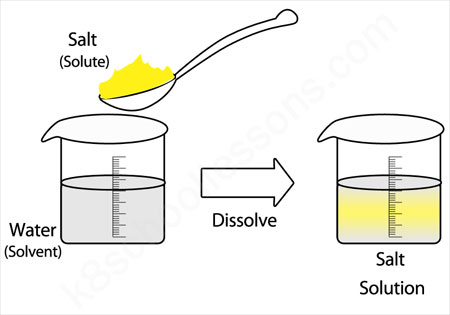


:max_bytes(150000):strip_icc()/how-to-make-saline-solution-608142_V2-01-5c8a85d1c9e77c0001a925f9.png)

:max_bytes(150000):strip_icc()/how-to-make-saline-solution-salt-water-mouth-rinse-4109216_final-b97b2e08087a41edb1c99758f5586207.png)


-6-187637.jpg)


