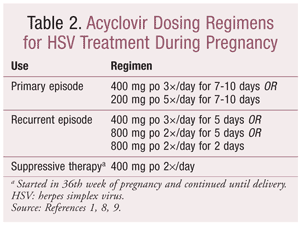
Effect of suppressive acyclovir administered to HSV-2 positive mothers from week 28 to 36 weeks of pregnancy on adverse obstetric outcomes: a double-blind randomised placebo-controlled trial | Reproductive Health | Full Text
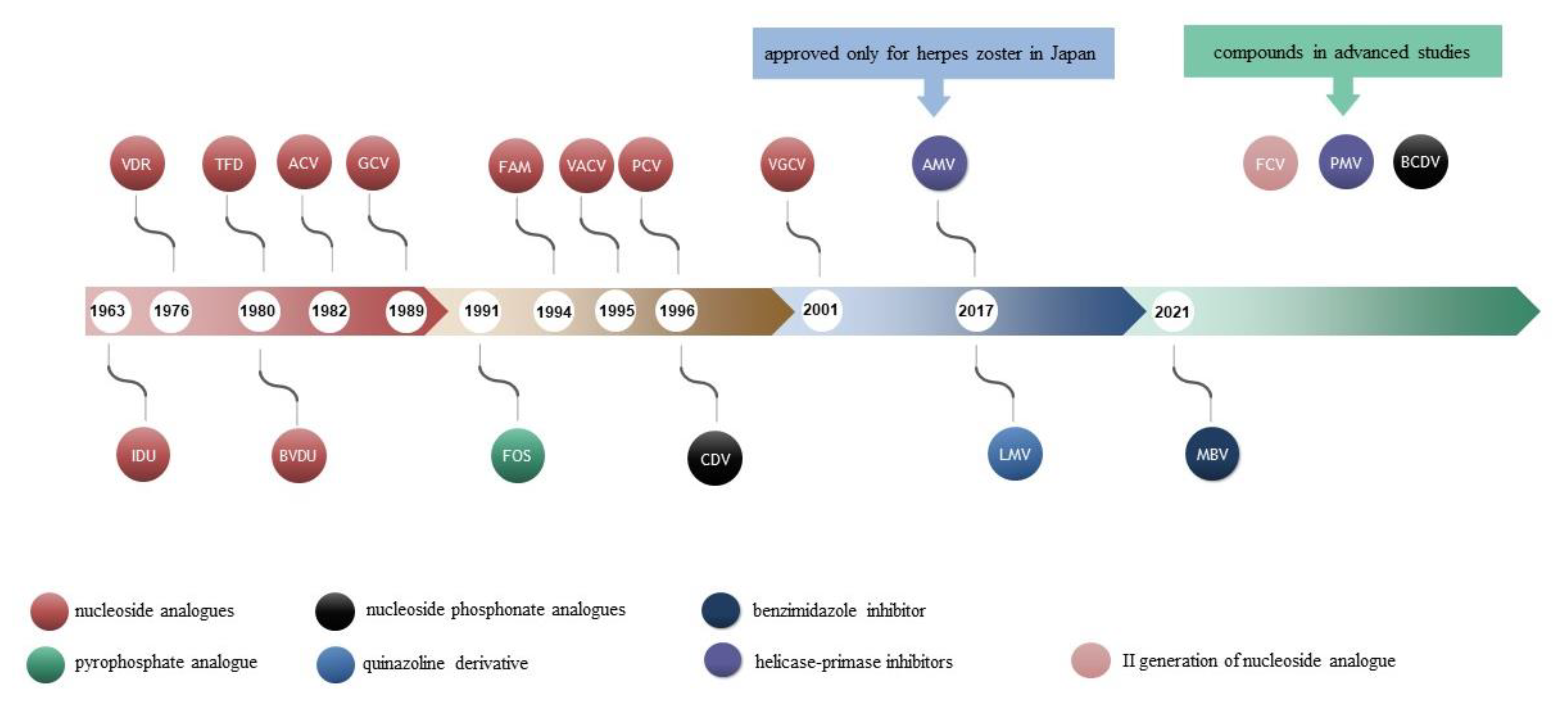
IJMS | Free Full-Text | 40 Years after the Registration of Acyclovir: Do We Need New Anti-Herpetic Drugs?







:max_bytes(150000):strip_icc()/pregnantwoman-ba6c8ba497ad4394b330ef70cedfd1b9.jpg)